What Is The Energy Required To Remove An Electron From An Atom
two.3.1: Ionization free energy
- Folio ID
- 198183
Ionization Energy
Ionization free energy (IE) is the free energy required to remove an electron from a neutral atom or cation in its gaseous phase. IE is also known as ionization potential.
\[A^{n+}_{(g)} \longrightarrow A^{(n+1)+}_{(g)} + due east^- \hspace{1cm} IE = \Delta U\]
Conceptually, ionization energy is the affinity of an element for its outermost electron (an electron information technology already has in its valence shell).
1st, twond, and iiird Ionization Energies
The symbol \(I_1\) stands for the first ionization free energy (energy required to take away an electron from a neutral cantlet, where \(n=0\)). The symbol \(I_2\) stands for the second ionization energy (energy required to take away an electron from an atom with a +1 charge, \(n=two\).)
Showtime Ionization Energy, \(I_1\) (general chemical element, A): \( A_{(g)} \rightarrow A^{1+}_{(yard)} + e^- \)
2d Ionization Energy, \(I_2\) (full general element, A): \( A^{i+}_{(m)} \rightarrow A^{two+}_{(g)} + due east^- \)
Tertiary Ionization Energy, \(I_3\) (general element, A): \( A^{2+}_{(one thousand)} \rightarrow A^{3+}_{(1000)} + e^- \)
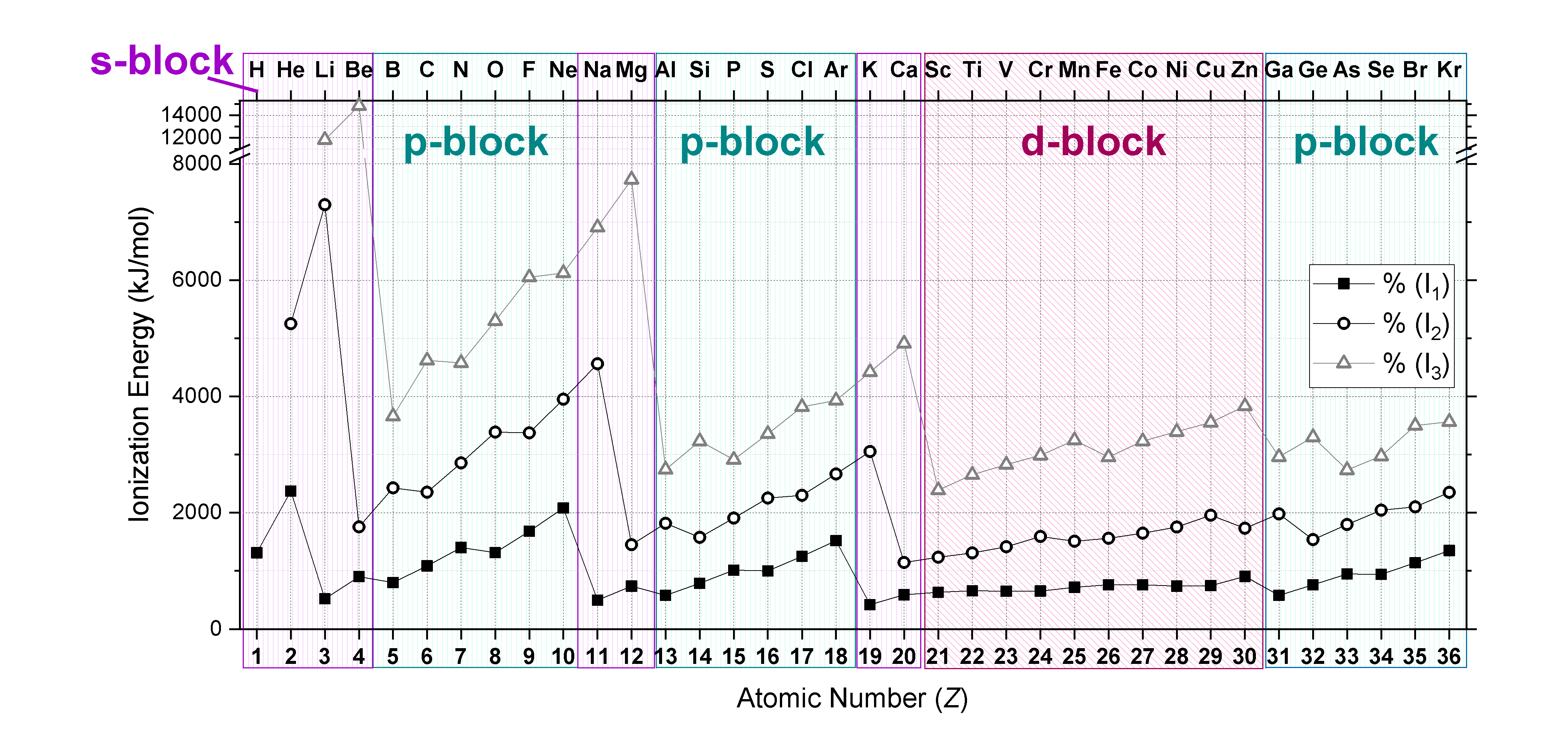
Each succeeding ionization energy is larger than the preceding energy. This means that \(I_1 < I_2 < I_3 < ... < I_n\) will e'er be truthful. For instance, ionization energy increases as succeeding electrons are taken away from Mg.
\[ Mg \,_{(grand)} \rightarrow Mg^+\,_{(yard)} + e^- \;\;\; I_1= 738\, kJ/mol \]
\[Mg^+ \,_{(g)} \rightarrow Mg^{2+}\,_{(g)} + e^- \;\;\; I_2= 1451\, kJ/mol\]
Ionization energy is correlated with the strength of attraction between the positively-charged nucleus and the negatively-charged valence electrons. The college the ionization energy, the stronger the attractive forcefulness betwixt nucleus and valence electrons, and the more energy is required to remove a valence electron. The lower the ionization free energy, the weaker the attractive force between nucleus and valence electrons, and the less free energy required to remove a valence electron.
Full general periodic trends in electron affinity
In general, ionization energies increment from left to correct and decrease down a group; however at that place are variations in these trends that would exist expected from the effects of penetration and shielding. The trends in first ionization free energy are shown in Figure \(\PageIndex{1}\) and are summarized below.
- Across a menstruum: As Z* increases beyond a period, the ionization energy of the elements generally increases from left to correct. However there are breaks or variation in the trends in the following cases:
- IE is peculiarly depression when removal of an electron creates a newly empty p subshell (examples include \(I_1\) of B, Al, Sc)
- IE energy is peculiarly low where removal of an electron results in a one-half-filled p or d subshell (examples include \(I_1\) of O, Due south)
- IE increases more gradually across the d- and f-subshells compared to due south- and p- subshells. This is because d- and f- electrons are weakly penetrating and feel peculiarly low Z*.
- From one period to the next: At that place is an specially large decrease in IE with the start of every new period (from He to Li or from Ne to Na for example). This is consequent with the idea that IE is especially low when removal of an electron creates a newly empty due south-subshell.
- Nobel gases: The noble gases posses very loftier ionization energies. Annotation that helium has the highest ionization energy of all the elements.
- Downwardly a group: Although Z* increases going down a grouping, there is no reliable trend in IE going down any group; in some cases IE increases going downwards a grouping, while in other cases IE decreases going downwards a group.
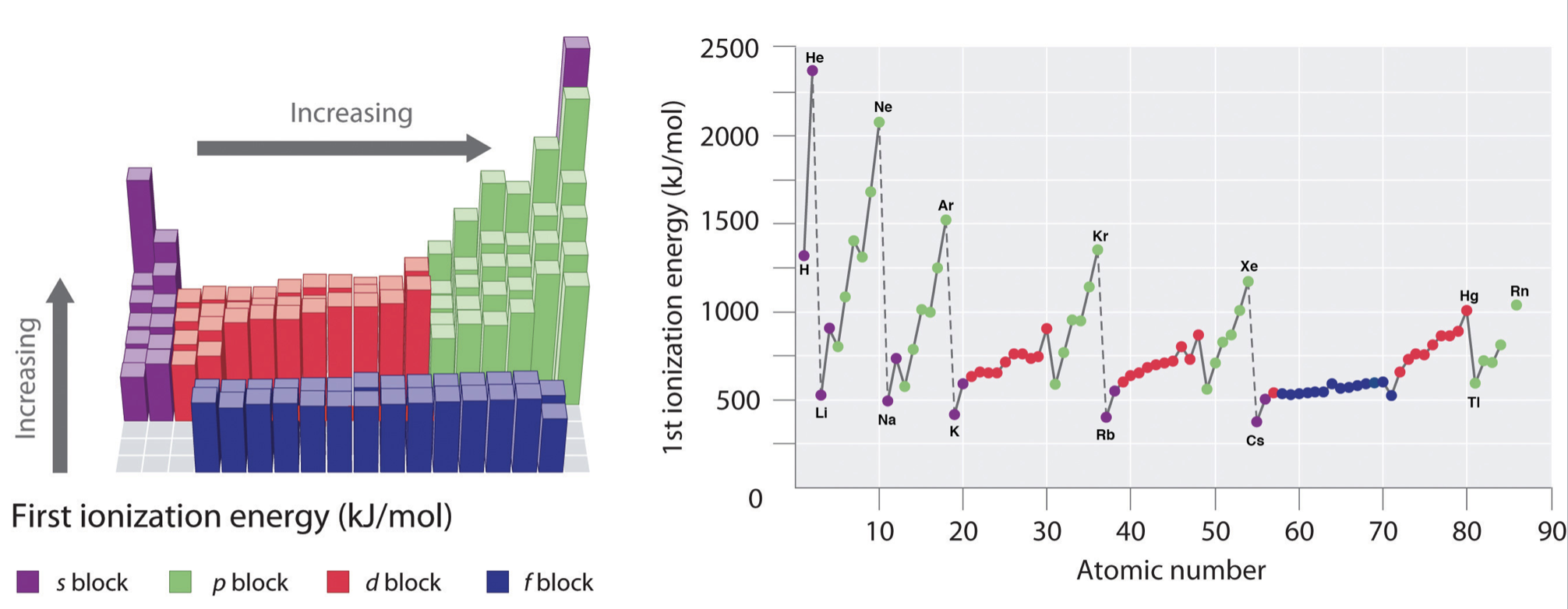
Figure \(\PageIndex{ane}\): Graph showing the First Ionization Energy of the Elements. Left: ionization energies are mapped onto the periodic tabular array, where the magnitude of ionization energy is depicted as a bar graph. Right: the starting time ionization energy is plotted confronting atomic number. In both panels the elements of the s-cake are shown in regal, p-block are shown in green, d-block are shown in reddish, and f-block are shown in blue.
Plots of the \(I_1, I_2,\) and \(I_3\) of elements from hydrogen to krypton (first four periods) are shown in Effigy \(\PageIndex{2}\). Notice that \(I_3>I_2>I_1\). Also notice that trends mentioned to a higher place for \(I_1\) concur true for subsequent ionizations when electron configurations are considered!
Contributors and Attributions
- Swetha Ramireddy (UCD), Bingyao Zheng (UCD), Emily Nguyen (UCD)
-
Modified or created by Kathryn Haas (khaaslab.com)
Source: https://chem.libretexts.org/Bookshelves/Inorganic_Chemistry/Map:_Inorganic_Chemistry_%28Miessler_Fischer_Tarr%29/02:_Atomic_Structure/2.03:_Periodic_Properties_of_Atoms/2.3.01:_Ionization_energy
Posted by: robertsgois1936.blogspot.com


0 Response to "What Is The Energy Required To Remove An Electron From An Atom"
Post a Comment